Principal Investigator
- Home
- Dr. Nandini Verma

Dr. Nandini Verma
Principal Investigator
Honors and Recognitions
- Assistant Professor, Homi Bhabha National Institute (HBNI), Mumbai.
- Prestigious Koshland Fellowship, Koshland Foundation, McDonald-Leapman Grant, 2017-18.
- Senior Post-Doctoral Fellowship for academic excellence, Feinberg Graduate School, Weizmann Institute of Science, 2016-17.
- Dean of Faculty post-doctoral fellowship, Weizmann Institute of science, 2012 -2014.
- VATAT-PBC Indo-Israeli Fellowships for outstanding post-doctoral researchers, 2012-2015.
- ISCA-Young Scientist Award, National Nominee, 2011.
- Foreign travel grant, CSIR, 2008.
- CSIR-JRF - NET fellowship in life sciences (12/2004).
- UGC-JRF - NET fellowship in life sciences (06/2004).
- University of Delhi, M.Sc. final topper (04/2004).
Research Interests
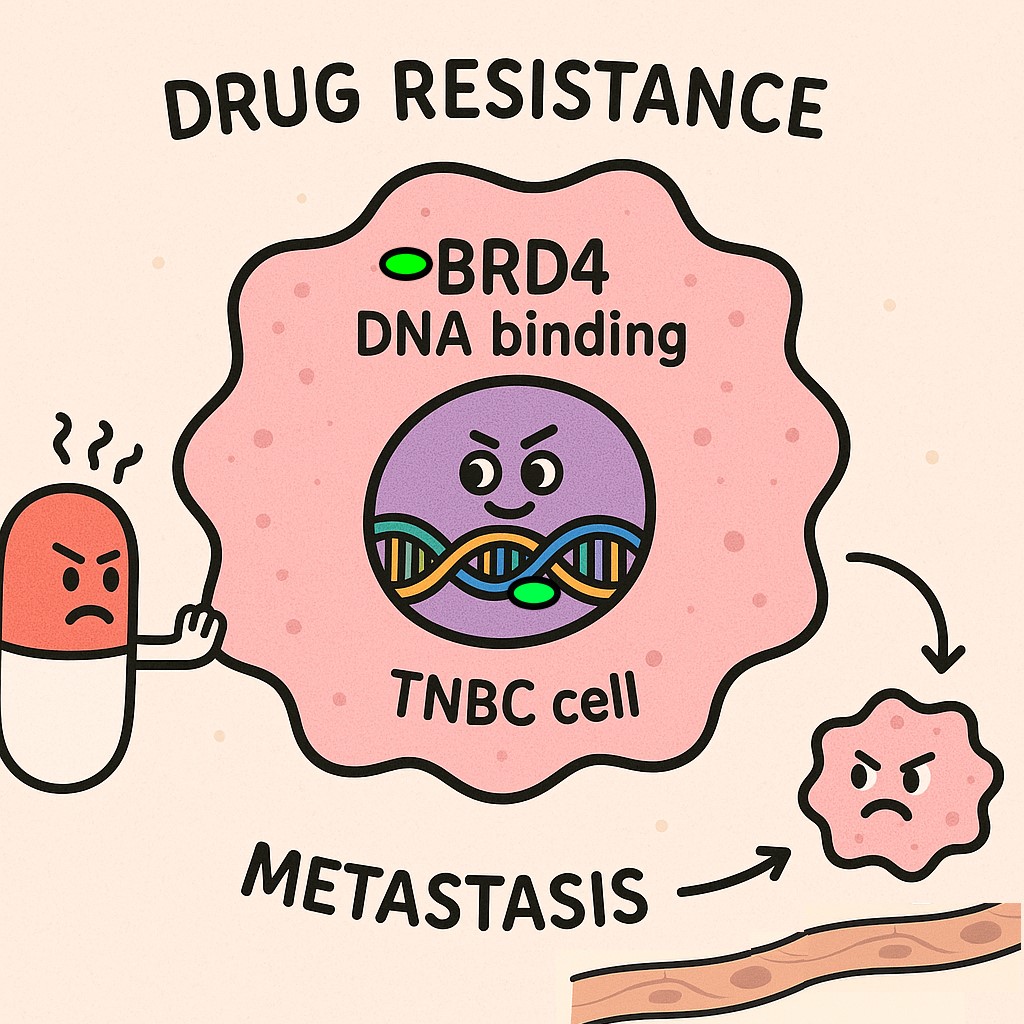
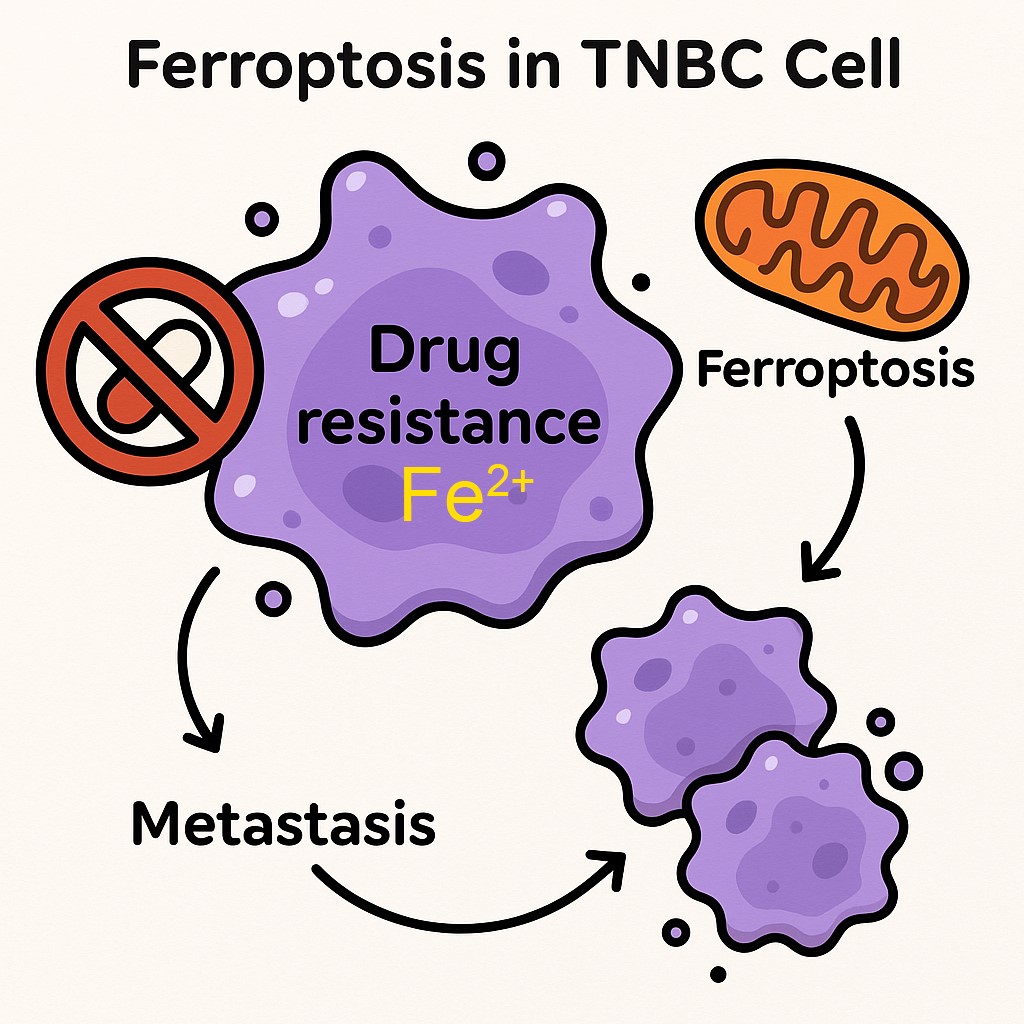
Drug-Tolerant persister (DTP) cancer cells are representative of residual disease in tumors post- therapy, these are the fraction of cells that undergo extensive molecular reprograming to show persister phenotypes, mainly resistance to apoptosis, EMT, and ferroptosis sensitivity.
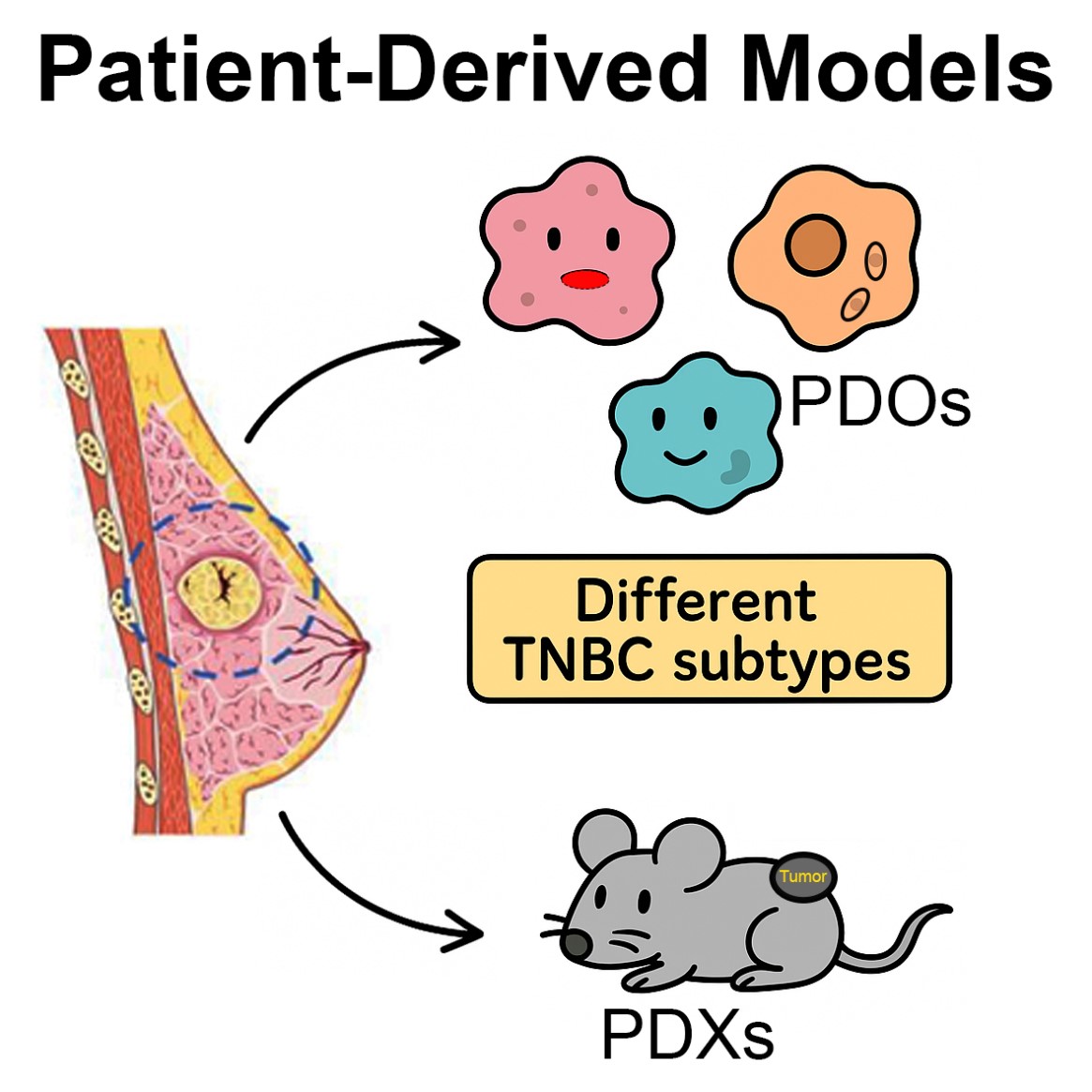
Our research group have developed several unique TNBC subtype specific cellular models for DTPS to major chemo- and targeted therapeutics available in clinic for TNBC treatment. These DTP models will facilitate the understanding of subtype specific molecular mechanisms involved in the TNBC DTP cell development and maintenance. These studies are being used to identify novel patho-physiological vulnerabilities unique to TNBC subtypes that can be used to design subtype specific precision medicine and can overcome drug resistance to TNBC clinically used therapies. Main focus is on the following specific topics.
- DTP cell specific molecular networks of extra- and intra-cellular signalling through cell surface receptors
- Cellular reactive oxygen species and heavy metal ions in drug tolerance and EMT
- Epigenetic factors governing drug tolerance and metastasis phenotypes of TNBC DTP cells
- Ferroptosis pathway molecules that can regulate TNBC DTP cell survival and maintenance
RESEARCH
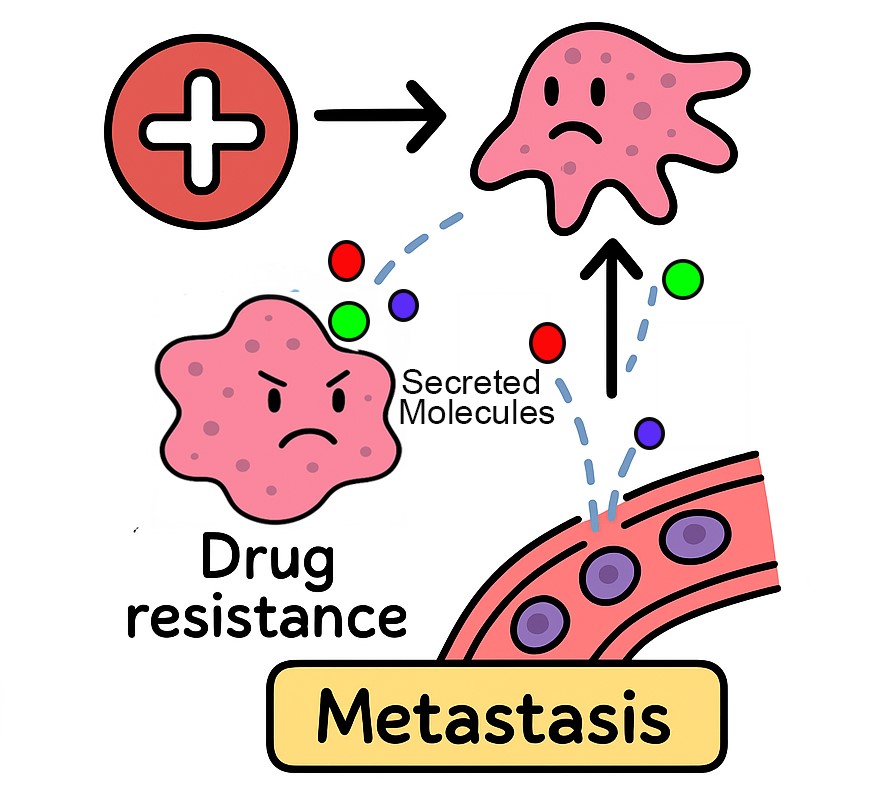
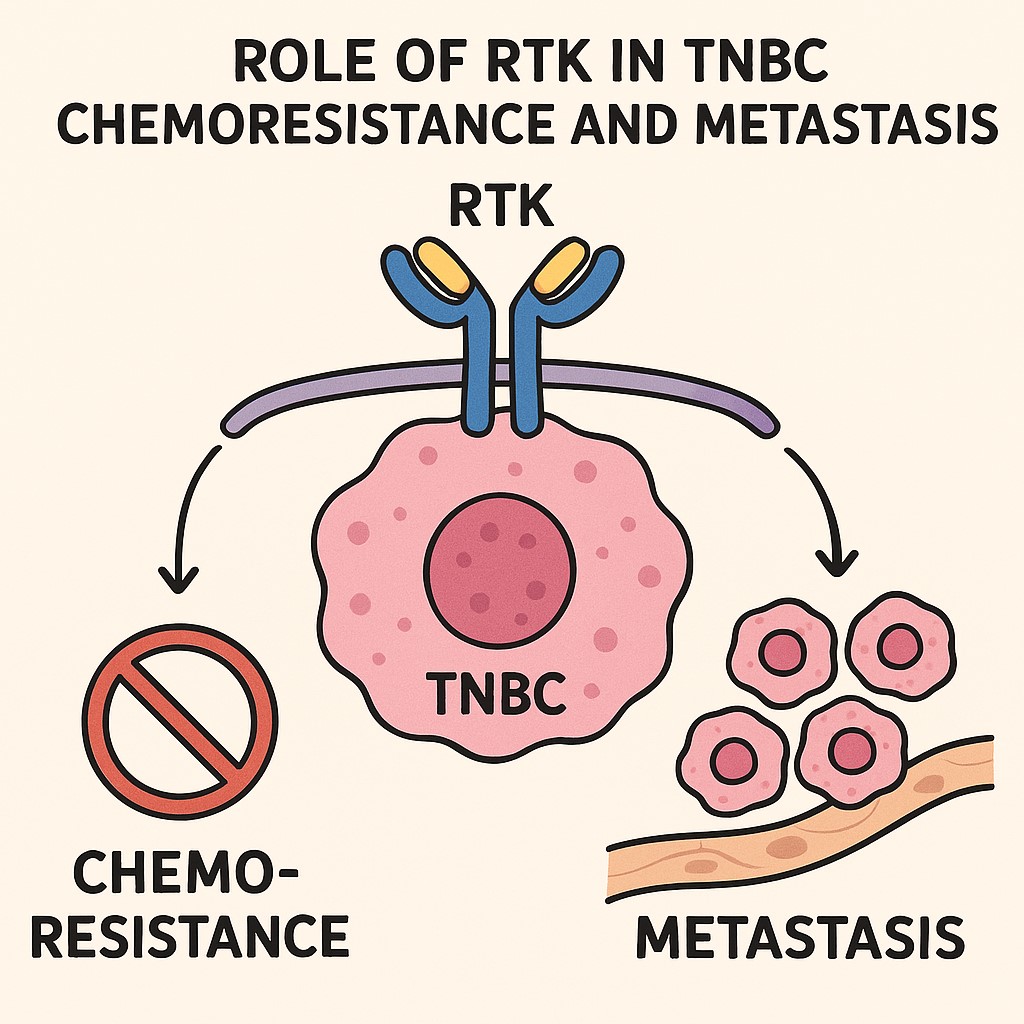
Triple Negative Breast cancer (TNBC) is a very heterogeneous, aggressive and metastatic subset of breast malignancy with worse prognosis and shortest disease-free survival rate. Pathologically, TNBC is defined by the lack of hormone receptors (estrogen, ER and progesterone, PR), as well as of human epidermal growth factor receptor2 (HER2) expression, however, it can be further classified into 4-6 distinct molecular subtypes. TNBC accounts for 15-25% of total breast cancers cases detected worldwide, however, recent clinical statistics from different regions of India revealed an excessive burden of TNBC among Indian women (up to a 31% mean prevalence) greatest as compared to any other regions of the world. As obvious targets like ER/PR and HER2 are absent in TNBC, standard clinical management relies mainly on chemotherapeutic agents in neoadjuvant setting or post-resection. Due to high proliferation index TNBC are chemo-sensitive and a fraction of patients (30-45%) does attain initial pathological complete response (pCR). However, several of these cases have early relapse (within 2-5 years post-therapy) with distant metastasis. Moreover, a significant number of patients (>50%) either fail to achieve pCR or have progressed disease during the therapy with metastasis. Management of relapsed chemo-resistant TNBC is extremely challenging and overall prognosis of such patients is often very poor due to limited treatment options, as both choice and response to other therapeutic options (targeted/combination-therapy) are often unpredictable at this stage. Our research laboratory is interested in understanding the molecular, epigenetic and secretome determinants in different subtypes of TNBC tumors that contribute towards the drug resistance to first-line clinical drugs in TNBC Patients.
Our research lab is focused on the critical questions that how different TNBC subtypes attain resistance to therapy and metastasis under treatment and how these molecular events can be intercepted? We use clinically driven sophisticated cellular and animal model systems of TNBC to study transcriptome, proteome, epigenome and secretome landscapes, together with cutting edge high-throughput screening approaches to answer these questions. These studies are expected to unmask several novel drug resistance mechanisms in specific TNBC subtypes and hence, can provide rationales for specific therapeutic strategies to re-sensitize cancer cells to cytotoxic therapies used in clinic. The ultimate goal of our studies will be to discover TNBC subtype specific precision medicine for Indian patients that can overcome drug resistance and minimize disease relapse, and thus, can improve the patient’s outcome and survival after therapy.